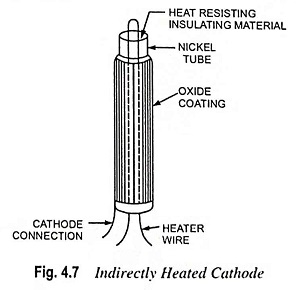
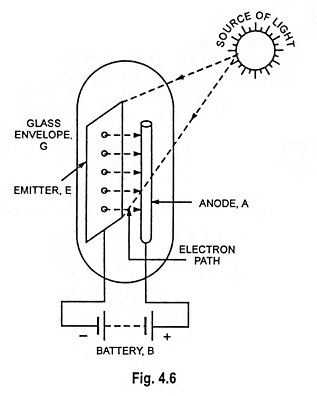
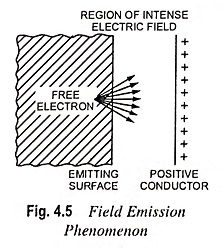
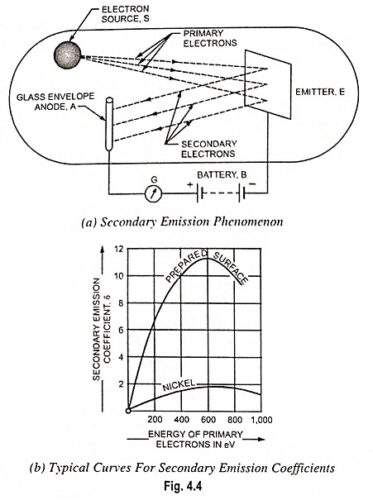
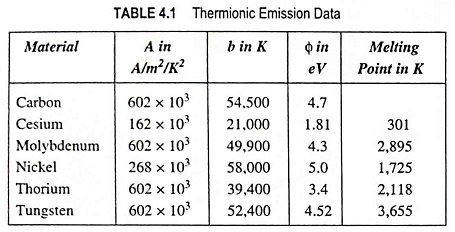
What is Cathode or Emitter and Types of Cathode?
What is Cathode or Emitter and Types of Cathode? The substance employed for electron emission is called a cathode or emitter. It is an essential part of an electron tube. The usefulness of a particular…